 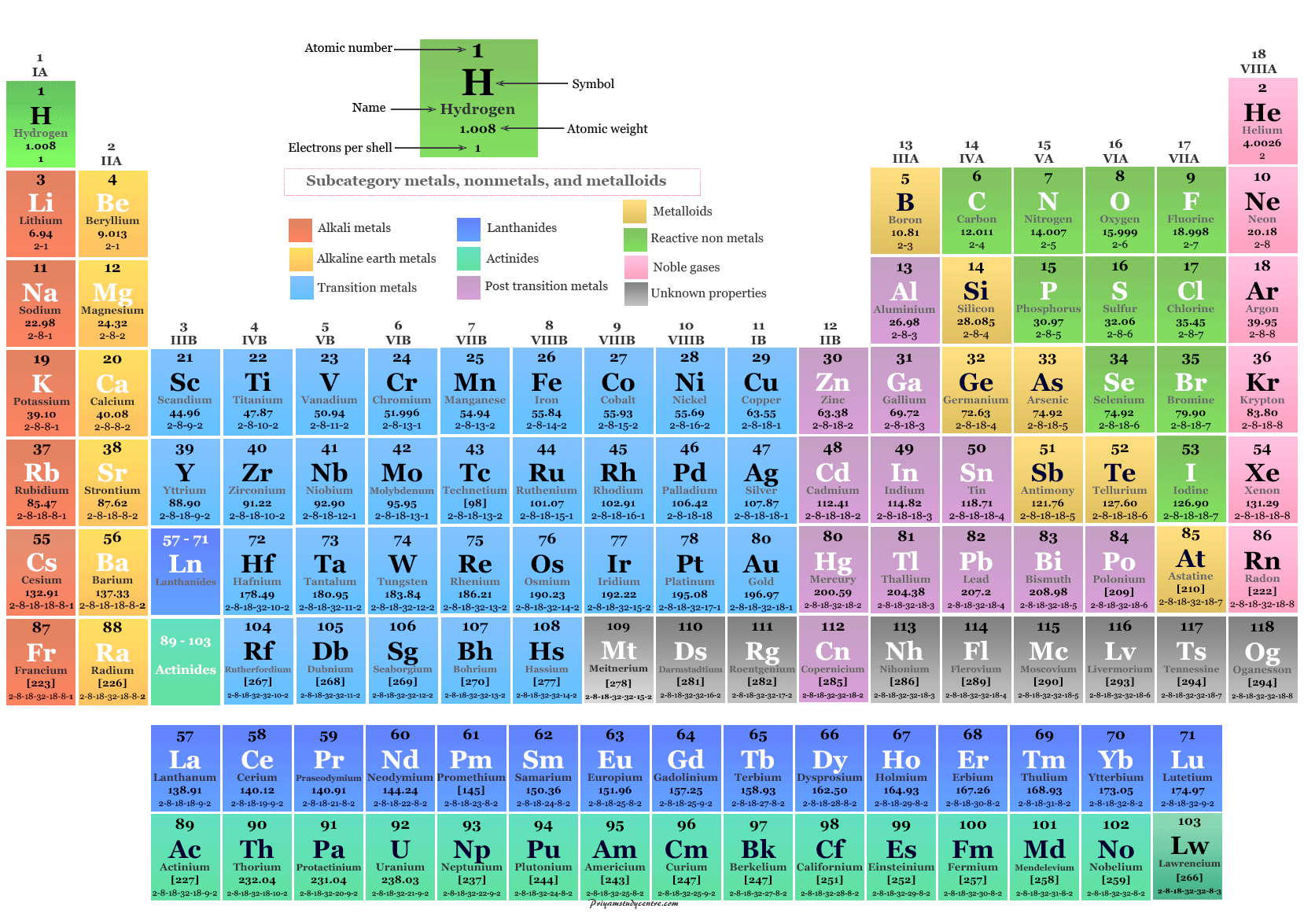
Between zinc (Zn) and arsenic (As) are two such missing elements. Notice that there are several places in the table that have no chemical symbol, but are instead labeled with a question mark. As we will see shortly, this turned out not to be the case, but Mendeleev was indeed correct to group these two elements as he did. He simply assumed that there was an error in the determination of one or both of the atomic masses. Mendeleev reversed the order because he knew that the properties of iodine were much more similar to those of fluorine (F), chlorine (Cl), and bromine (Br) than they were to oxygen (O), sulfur (S), and selenium (Se). One of Mendeleev’s insights is illustrated by the elements tellurium (Te) and iodine (I). Notice that tellurium is listed before iodine even though its atomic mass is higher. As a result, elements that are in the same horizontal row are groups of elements that were known to exhibit similar chemical properties. In Figure 2, atomic mass increases from top to bottom of vertical columns, with successive columns going left to right. It was not until years later that another more extensive periodic table effort would gain much greater acceptance and the pioneering work of John Newlands would be appreciated.įigure 2. Mendeleev’s 1869 periodic table. 
Newlands’s work was largely ignored and even ridiculed by the scientific community in his day. ” Unfortunately, there were some elements that were missing and the law did not seem to hold for elements that were heavier than calcium. He called this relationship the “Law of Octaves. While Dobereiner’s system would pave the way for future ideas, a limitation of the triad system was that not all of the known elements could be classified in this way.Įnglish chemist John Newlands (1838-1898) ordered the elements in increasing order of atomic mass and noticed that every eighth element exhibited similar properties. These three elements also displayed similar chemical reactions, such as vigorously reacting with the members of another triad: chlorine, bromine, and iodine. When the atomic masses of lithium and potassium were averaged together, it was approximately equal to the atomic mass of sodium (22.99). Dobereiner found that the atomic masses of these three elements, as well as other triads, formed a pattern. Triads were based on both physical as well as chemical properties. One such triad was lithium, sodium, and potassium. In 1829, a German chemist, Johann Dobereiner (1780-1849), placed various groups of three elements into groups called triads. In other words, putting elements in separate groups based on how they reacted with other elements. The question was: how?Ī logical way to begin grouping elements together was by their chemical properties. With the ever-increasing number of elements, chemists recognized that there may be some kind of systematic way to organize the elements. As scientific methods improved, the rate of discovery dramatically increased. Several of these, such as copper and lead, had been known since ancient times. Early Attempts to Organize Elementsīy the year 1700, only a handful of elements had been identified and isolated. Chemistry information also needs to be organized so we can see patterns of properties in elements. Both approaches organize information so that people can easily find what they are looking for. This method is one of the most widely used ways to organize libraries in the world. This number refers to the Dewey Decimal system, developed by Melvil Dewey in 1876 and used in over 200,000 libraries throughout the world.Īnother system in wide use is the Library of Congress approach, developed in the late 1800s-early 1900s to organize the materials in the federal Library of Congress. The book you are looking for will have a number by the title. If you are looking for a non-fiction publication, you look in a catalog (most likely on a computer these days). 
If it is a fiction book, you look by author since the fiction materials are filed by the author’s last name. When you go to the library to find a book, how do you locate it? 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
